- 1. Indicate if the following statements are TRUE (T) or FALSE (F) about acetylcholine (ACh) and cholinergic neurotransmission.
- a. (FALSE) AChE [CAT]is the enzyme that catalyzes the condensation of choline and acetate in the biosynthesis of Ach.
- b. (FALSE) Muscarinic ACh receptors are G-protein linked sites involving the generation of the second messenger cyclic AMP [DAG and IP3] through the catalytic action of adenylyl cyclase [phospholipase C].
- c. (TRUE) ACh is a chemically reactive molecule that readily undergoes hydrolysis to choline and acetate in aqueous solution.
- d. (FALSE) The plasma tẄ of ACh is relatively short due to its rapid catabolism by the enzyme choline acEtyltransferase (CAT) [esterases].
- e. (FALSE) ACh activation of muscarinic AChRs results in an increase in intracellular levels of inositol triphosphate which, in turn, triggers the activation of the enzyme phosphokinase C [protein kinase C].
- f. (FALSE) Activation of niotinic AChRs results in increased efflux of Ca++ [Na+ and Cl-] through receptor-operated membrane channels.
- g. (FALSE) Muscarinic AChR selectivity of ACh can be increased by addition of an alpha [ß]-CH3 structural feature to choline esters.
- h. (TRUE) A carbamate derivative of ACh, e.g. H2NCOOCH2CH2N(CH3)3+, would be expected to possess a longer plasma tẄ than ACh.
- i. (TRUE) Direct-acting cholinomimetics possess miotic activity when administered intraocularly.
- j. (FALSE) Diacylglycerol is a second-messenger of ACh in cells innervated by MAChRs and is involved in triggering the release of Ca++ [activating protein kinase C].
- b. (FALSE) Muscarinic ACh receptors are G-protein linked sites involving the generation of the second messenger cyclic AMP [DAG and IP3] through the catalytic action of adenylyl cyclase [phospholipase C].
- 2. The following questions refer to to the graphical depiction of the pH dependency of AChE inhibition by physostigmine and neostigmine (Figure 2).

- a. Which drug, physostigmine or neostigmine, shows a dependency on pH for its inhibitory activity? Explain why?
Because of the need to possess a cationic N for effective interaction with the anionic site of AChE, physostigmine, a tertiary amine, will exhibit pH dependency in its inhibitory effects on AChE, i.e, greatest inhibitory activity at acidic pH where it is largely cationized.
- b Which AChEI, physostigmine or neostigmine, shows no dependence on pH for its inhibitiory activity (line B)? Explain why?
Neostigmine, a quaternary ammonium salt, will be fully cationized regardless of pH and will therefore be able to effectively bind to AChE regardless of the pH of the medium.
- c. Which drug, neostigmine or physostigmine, would be preferred in treating Alzheimer's disease? Explain why.
Because it is a tertiary amine and will better cross the blood-brain barrier, physostigmine would be preferred in the treatment of the CNS disorder, Alzheimer's Disease, compared to neostigmine which will NOT be distributed well to the CNS.
- Explain why the analogue of neostigmine shown on the structure page (STRUCTURE 2d) is a much less potent AchEI.
- b Which AChEI, physostigmine or neostigmine, shows no dependence on pH for its inhibitiory activity (line B)? Explain why?
The analogue structure, having a 1,4-relationship between the key carbamylating and quaternary ammonium functions, will not "fit" well at the active site of AChE and will therefore produce only weak inhibition.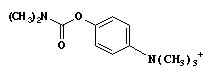
- 3. Complete the following reaction by showing the structure of the complex formed between edrophonium and the active site of AChE.

- a. Would you expect the interaction between edrophonium and AChE to be reversible or irreversible? Explain your answer.
As shown, edrophonium establishes bioreversible H-bonds and ionic bonds with the active site groups of AChE as opposed to highly stable covalent bonds. Therefore this constitutes a reversible (competitive) interaction with the enzyme.
- b. Would you expect edrophonium to be a substrate or an inhibitor of AChE? Explain your answer.
Edrophonium has no functional groups subject to catalytic hydrolysis by AChE, i.e., this structure CANNOT function as a substrate but, rather, as an inhibitor.
- b. Would you expect edrophonium to be a substrate or an inhibitor of AChE? Explain your answer.
- a. (FALSE) AChE [CAT]is the enzyme that catalyzes the condensation of choline and acetate in the biosynthesis of Ach.
 Return to:
Return to: