Med. Chem. II Weekly Review
and Study Guide
- A. Noradrenergic neurotransmission in the peripheral nervous system
- 1. Life Cycle of a Chemical Neurotransmitter (CNT)
- a. Biosynthesis
- b. Storage (vesicles)
- c. Release (Ca2+, exocytosis)
- d. Receptors (biochemistry of signal transduction)
- e. Termination
- 1) Uptake
- 2) Catabolism
- b. Storage (vesicles)
- 2. Physicochemical properties of norepinephrine
- a. General structure: ß-arylethanolamine
- b. Acid-base properties:
- 1) NE is a strong organic base (NH2) and a weak organic acid (catechol -OH)
- 2) Cationic at physiologic pH
- 3) Forms salts having greater H2O solubility upon reaction with acids
- 2) Cationic at physiologic pH
- c. Chiral molecule: R enantiomer is naturally-occurring, physiologically-active isomer.
- d. Catechol relatively easily oxidized to quinone (bioinactive) in air.
- b. Acid-base properties:
- 3. Structure and activity of norepinephrine as an adrenoceptor ligand
- a. Biosynthesis
- B. Drugs Affecting Adrenergic Neurotransmission at Presynaptic Sites
- 1. Drugs affecting NE biosynthesis (click here for diagram)
PRACTICE QUESTIONS
[Answers in red]
- 1. What is the structural classfication of NE? [ß-arylethanolamine derivative]. How does the structure of NE differ from that of the other adrenergic neurotransmitters/neuromodulators, dopamine and epinephrine? [ß-OH + primary NH2]. Which of these three catecholamines are chiral? [Those with a ß-OH function]. Is chirality important in the physiological actions of these catecholamines? [Yes, the various ARs are highly stereoselective in their interaction with optical isomers of the catecholamines, preferring the R-configuration.]
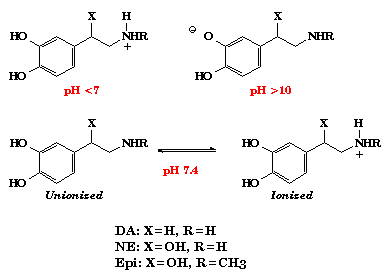
- 2. Draw the structures of NE, DA and EPI as these catecholamines exist at (a) acidic pH (<7), (b) physiologic pH and (c) alkaline pH (>10).
- 3. NE has been proposed to interact with adrenergic receptors via a 3-point attachment utilizing functional groups in the aryl, side chain and amine portions of its structure. List the types of attractive forces that will likely be established between NE and its receptor surface.
- Catechol -OH's can form H-bonds with complementary sites on AR surface.
ß-OH can similarly form H-bonds with acceptor/donor sites at AR sites.
cationic ammonium group (at phys. pH) can ionically interact with an anionic site on receptor.
- 4. Indicate if the following drug effects on NE neurotransmission will result in an increase, a decrease, both or no change in overal rate of neurotransmission. Explain your answers.
- a. Displacement of NE from presynaptic stores - [mimetic effect if displacement mimics nerve impulse, e.g. indirect-acting ß-arylethylamines and acute actions of adrenergic neuronal blockers or lytic effect if displacement results in loss of NE for neurotransmission process]
- b. Stabilization (prevention of dissociation) of the heterotrimeric structure of the ß-adrenoceptor Gs regulatory protein [ß-adrenolytic action by interfering with the receptor transduction process].
- c. Inhibition of tyrosine hydroxylase [adrenolysis due to diminished concentrations of NE in adrenergic nerve ending].
- d. Inhibition of phospholipase C in alpha-adrenergically-innervated effector tissue [alpha-adrenolytic action by interfering with the receptor transduction process].
- e. Competitive antagonism of NE at presynaptic alpha2-adrenoceptors [alpha2-sympathomimetic effect by interfering with regulation of NE release from nerve ending].
- 5. Draw the reactions involved in the biosynthesis of NE and explain the role of the following enzyme cofactors in this biosynthesis.
- a. Tetrahydrobiopterin for tyrosine hydroxylase - used to generate oxygen radical required for insertion into C-H bond at 3-position of tyrosine to yield L-DOPA.
- b. Pyridoxal phosphate for L-AAAD - used by enzyme to form condensation product with L-DOPA which facilitates decarboxylation to yield dopamine.
- c. Ascorbic acid for dopamine ß-hydroxylase - - used to generate oxygen radical required for insertion into C-H bond at ß-position of dopamine to yield R-NE.
- 6. Show the primary extraneuronal and neuronal catabolic pathways for NE. List the enzymes involved in catalyzing each reaction.
Refer to page 11 of Lecture Guide and Web diagram
- 7. Comment on the role of each of the following parts of the NE on (i) receptor activity, (ii) relative solubility, (iii) metabolic stability and (iv) chemical stability:
- a. the amino group
- b. the ethyl chain connecting the amino and aryl groups
- c. the catechol structure.
Check this diagram (click HERE)
- 1. What is the structural classfication of NE? [ß-arylethanolamine derivative]. How does the structure of NE differ from that of the other adrenergic neurotransmitters/neuromodulators, dopamine and epinephrine? [ß-OH + primary NH2]. Which of these three catecholamines are chiral? [Those with a ß-OH function]. Is chirality important in the physiological actions of these catecholamines? [Yes, the various ARs are highly stereoselective in their interaction with optical isomers of the catecholamines, preferring the R-configuration.]
- 2. Draw the structures of NE, DA and EPI as these catecholamines exist at (a) acidic pH (<7), (b) physiologic pH and (c) alkaline pH (>10).

- 3. NE has been proposed to interact with adrenergic receptors via a 3-point attachment utilizing functional groups in the aryl, side chain and amine portions of its structure. List the types of attractive forces that will likely be established between NE and its receptor surface.
- Catechol -OH's can form H-bonds with complementary sites on AR surface.
ß-OH can similarly form H-bonds with acceptor/donor sites at AR sites.
cationic ammonium group (at phys. pH) can ionically interact with an anionic site on receptor.
- 4. Indicate if the following drug effects on NE neurotransmission will result in an increase, a decrease, both or no change in overal rate of neurotransmission. Explain your answers.
- a. Displacement of NE from presynaptic stores - [mimetic effect if displacement mimics nerve impulse, e.g. indirect-acting ß-arylethylamines and acute actions of adrenergic neuronal blockers or lytic effect if displacement results in loss of NE for neurotransmission process]
- b. Stabilization (prevention of dissociation) of the heterotrimeric structure of the ß-adrenoceptor Gs regulatory protein [ß-adrenolytic action by interfering with the receptor transduction process].
- c. Inhibition of tyrosine hydroxylase [adrenolysis due to diminished concentrations of NE in adrenergic nerve ending].
- d. Inhibition of phospholipase C in alpha-adrenergically-innervated effector tissue [alpha-adrenolytic action by interfering with the receptor transduction process].
- e. Competitive antagonism of NE at presynaptic alpha2-adrenoceptors [alpha2-sympathomimetic effect by interfering with regulation of NE release from nerve ending].
- b. Stabilization (prevention of dissociation) of the heterotrimeric structure of the ß-adrenoceptor Gs regulatory protein [ß-adrenolytic action by interfering with the receptor transduction process].
- 5. Draw the reactions involved in the biosynthesis of NE and explain the role of the following enzyme cofactors in this biosynthesis.
- a. Tetrahydrobiopterin for tyrosine hydroxylase - used to generate oxygen radical required for insertion into C-H bond at 3-position of tyrosine to yield L-DOPA.
- b. Pyridoxal phosphate for L-AAAD - used by enzyme to form condensation product with L-DOPA which facilitates decarboxylation to yield dopamine.
- c. Ascorbic acid for dopamine ß-hydroxylase - - used to generate oxygen radical required for insertion into C-H bond at ß-position of dopamine to yield R-NE.
- b. Pyridoxal phosphate for L-AAAD - used by enzyme to form condensation product with L-DOPA which facilitates decarboxylation to yield dopamine.
- 6. Show the primary extraneuronal and neuronal catabolic pathways for NE. List the enzymes involved in catalyzing each reaction.
Refer to page 11 of Lecture Guide and Web diagram - 7. Comment on the role of each of the following parts of the NE on (i) receptor activity, (ii) relative solubility, (iii) metabolic stability and (iv) chemical stability:
- a. the amino group
- b. the ethyl chain connecting the amino and aryl groups
- c. the catechol structure.
Check this diagram (click HERE) - b. the ethyl chain connecting the amino and aryl groups
- 2. Draw the structures of NE, DA and EPI as these catecholamines exist at (a) acidic pH (<7), (b) physiologic pH and (c) alkaline pH (>10).
 Return to:
PY421 Home Page
Return to:
PY421 Home Page