Med. Chem. II Week Two
Review and Study Guide
- Drugs Affecting Presynaptic/Prejunctional Steps of NE Neurotransmission
- 1. Drugs affecting NE biosynthesis
- 2. Drugs affecting NE storage and/or release
- a. Adrenergic neuronal blockers - displace stored NE and prevent restorage (chronic effect - sympatholytic effect)
- 1) General structural appearance (guanidines, quaternary ammonium salts)
- 2) Cationization (pharmaceutical and pharmacological importance)
- b. Indirect-acting sympathomimetics - displace stored NE similar to neuronal release (chronic effect - sympathomimetic effect)
- 1) ß-Arylethylamine structure vs. mode of sympathomimetic action
- 2) Physicochemical properties - basicity
- 1) General structural appearance (guanidines, quaternary ammonium salts)
3. Drugs affecting termination of NE neurotransmission (limited peripheral utility)
- a. Reuptake inhibitors (e.g. centrally-acting antidepressants)
- b. Catabolic enzyme inhibitors
- 1) MAOIs
- 2) COMT inhibitors
- b. Catabolic enzyme inhibitors
- 2. Drugs affecting NE storage and/or release
- 1. Alpha-adrenoceptor Agonists
- a. alpha1-AR Agonists
- 1) ß-Arylethylamines - structure-activity relationships; Prodrugs - mode of action
- b. alpha2-AR Agonists
- 1) Alpha-methyldopa (physicochemical properties, Mechanism of action, H2O solubilization
- 2) Imidazolines and Guanidines
- a) Structural considerations (resonance forms, role of bulky, lipophilic portion of structure in (i) receptor affinity and (ii) distribution to sites of action (CNS vs. PNS)
- b) Physicochemical properties (basicity, effect of base strength on (i) mode of action at adrenergic sites and (ii) differential distribution to site of alpha2-AR receptors, dosage formulation, etc.)
- b) Physicochemical properties (basicity, effect of base strength on (i) mode of action at adrenergic sites and (ii) differential distribution to site of alpha2-AR receptors, dosage formulation, etc.)
- 2) Imidazolines and Guanidines
- a. alpha1-AR Agonists
Click on highlighted, underlined text for diagrams and more info.
| Click on highlighted, underlined text for diagrams and more info. |
B. Drugs Affecting Adrenergic Receptor (AR) Functions
PRACTICE QUESTIONS
{Answers in italicized RED}
{Answers in italicized RED}
- 1. List two effects that alpha-methyldopa has on adrenergic neurotransmission.
{(1) inhibition of L-AAAD and (2) metabolic conversion to the potent presynaptic alpha2-agonist, alpha-methylNE}
- Which effect is most relevant to its therapeutic use?
{formation of the alpha2-agonist, alpha-methylNE}
- Explain the reason for using a prodrug of alpha-methyldopa for parenteral dosage formulation.
{alpha-MD exists as a low H2O soluble zwitterion in solution. Conversion to the methyl ester prodrug allows formulation of the drug as the H2O soluble HCl salt.}
- 2. Describe the physicochemical differences that exist between guanidine derivatives that are adrenergic neuronal blockers
{strong organic bases} and those that are alpha2-adrenergic receptor (AR) agonists {weaker organic bases}.
- Explain why each group has a different mechanism of adrenergic action.
{The strongly cationic (at phys. pH) ANB guanidines have high affinity for NE granules thereby depleting prejunctional NE stores while the less ionic alpha2-blocking guanidines can access the CNS and act at presynaptic sites to diminish NE neurotransmission.}
- List a common therapeutic use for each group of guanidine derivatives.
{management of mild to moderate hypertension}
- 3. Describe structural differences between a direct-acting and an indirect-acting sympathomimetic ß-arylethylamine derivatives.
{The direct-acting sympathomimetics have structural features more like NE itself including aromatic and side chain OH functions; the indirect-acting agents are more lipophilic having fewer OH functions and, perhaps, an alpha-CH3 substituent.}
- Which class of sympathomimetic will have higher AR-selectivity and why?
{The direct-acting agents because they act postjunctionally.}
- 4. Describe how the dichlorophenyl structural feature of clonidine effects its (i) AR affinity {alter the shape of this imidazoline derivative so that it best "fits" the NE-binding site at the AR} and (ii) site of action.
{lowers the basicity of the imidazoline structure increasing the conc. of unionized species plus adds to the lipid solubility of this species which aids the drug in accessing the CNS}.
- List and explain two reasons for the differences in therapeutic use of apraclonidine and clonidine.
{Apraclonidine, because of its more polar structure, (1) is not well distributed to the CNS and (2) its topical (ophthalmic) site of administration facilitates its use in the treatment of glaucoma}.
- 5. A recent report in the Journal of the American Medical Asssociation on the efficacy of midodrine in treating neurogenic orthostatic hypotension contained the statement that midodrine "is almost completely absorbed following oral administration and undergoes enzymatic hydrolysis to its active metabolite, desglymidodrine, with a bioavailability as desglymidodrine of 93%".
Show the reactions involved in converting midodrine to its active metabolite {refer to Lecture Guide for reaction} and comment on the pharmacologic and therapeutic significance of this process {desglymidodrine, because of first-pass extraction and metabolism, has low oral bioavailability while, its prodrug, midodrine avoids this site of loss thereby delivering therapeutic concentrations of the active drug systemically}.
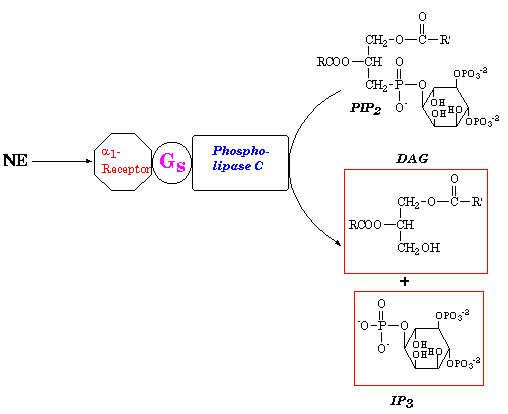
- 6. Complete the following scheme by filling the boxes with the appropriate structures for the products of this reaction.
{(1) inhibition of L-AAAD and (2) metabolic conversion to the potent presynaptic alpha2-agonist, alpha-methylNE}
- Which effect is most relevant to its therapeutic use?
{formation of the alpha2-agonist, alpha-methylNE}
- Explain the reason for using a prodrug of alpha-methyldopa for parenteral dosage formulation. {alpha-MD exists as a low H2O soluble zwitterion in solution. Conversion to the methyl ester prodrug allows formulation of the drug as the H2O soluble HCl salt.}
- Explain why each group has a different mechanism of adrenergic action.
{The strongly cationic (at phys. pH) ANB guanidines have high affinity for NE granules thereby depleting prejunctional NE stores while the less ionic alpha2-blocking guanidines can access the CNS and act at presynaptic sites to diminish NE neurotransmission.}
- List a common therapeutic use for each group of guanidine derivatives.
{management of mild to moderate hypertension}
{The direct-acting sympathomimetics have structural features more like NE itself including aromatic and side chain OH functions; the indirect-acting agents are more lipophilic having fewer OH functions and, perhaps, an alpha-CH3 substituent.}
- Which class of sympathomimetic will have higher AR-selectivity and why?
{The direct-acting agents because they act postjunctionally.}
{lowers the basicity of the imidazoline structure increasing the conc. of unionized species plus adds to the lipid solubility of this species which aids the drug in accessing the CNS}.
- List and explain two reasons for the differences in therapeutic use of apraclonidine and clonidine.
{Apraclonidine, because of its more polar structure, (1) is not well distributed to the CNS and (2) its topical (ophthalmic) site of administration facilitates its use in the treatment of glaucoma}.
Show the reactions involved in converting midodrine to its active metabolite {refer to Lecture Guide for reaction} and comment on the pharmacologic and therapeutic significance of this process {desglymidodrine, because of first-pass extraction and metabolism, has low oral bioavailability while, its prodrug, midodrine avoids this site of loss thereby delivering therapeutic concentrations of the active drug systemically}.

 Return to:
PY421 Home Page
Return to:
PY421 Home Page